The equation of all equations:
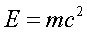
Everyone has seen this equation, and everyone
knows this is
Einstein's famous "mass equals energy"
equation.
But what does it mean exactly? As always, its
best to see it in an example. Because
Einstein's
work was with
light and energy, the best example is
the
Sun. Our
Sun produces energy through fusion -
the combining of 4 hydrogen
atoms to make a helium
atom plus a few extra parts. When comparing the mass
of 4 hydrogen
atoms to a helium
atom, there is some
mass loss:
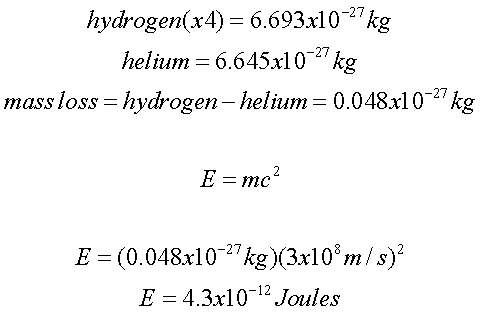
Keep in mind that this is EACH reaction. In the
Sun, there are millions of these reactions
occurring.
Back to Top
|

